Bacterial co-infections in COVID-19 patients: what consequences for the clinical picture and treatment?
Bacterial co-infections in patients with COVID-19: the article “When two for the price of one isn’t a bargain: estimating prevalence and microbiology of bacterial co-infections in patients with COVID-19” (PDF at the end), from which a part of the title has been taken, leads to right reflections on the topic of co-infections and COVID-19 that should be deepened and suggests the reading of a literature review on the subject.
The prevalence and microbiology of concomitant bacterial infections in patients with SARS-CoV-2 infection are not yet well known.
We know that in people with viral respiratory tract infections, the presence of a concomitant bacterial infection has been associated with poor clinical results.
For example, in influenza patients, an overlapping bacterial infection is present in 20-30% of patients and has been associated with increased shock, mechanical ventilation, and mortality rates.
Similarly, in children with severe respiratory syncytial virus (RSV) infection, multiple studies have shown overlapping bacterial pneumonia rates over 30%, and this has been associated with increased duration of mechanical ventilation.
Studying the rates and microbiology of bacterial co-infection in patients with viral respiratory infections can help determine empirical antibiotic therapy, understand the prognosis and discern pathogenesis in viral-bacterial co-infections.
Coinfections and COVID-19: a “living rapid review” with meta-analysis for continuous update
Living Reviews are reviews that are continuously updated by incorporating relevant new evidence as it becomes available.
Langford and his colleagues conducted a living rapid review (“Bacterial co-infection and secondary infection in patients with COVID-19: a living rapid review and meta-analysis” / PDF) of studies that examined the rates of bacterial pneumonia or blood flow infection in patients with COVID-19.
The studies were included in the review if:
- they evaluated patients with confirmed COVID-19 and
- have reported the prevalence of acute bacterial infection.
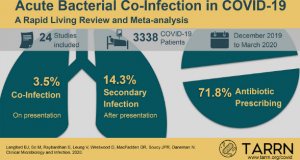
Of the 1308 studies initially selected, 24 were eligible and included in the review, for a total of 3338 patients with COVID-19 assessed for acute bacterial infection.
In the meta-analysis, bacterial co-infection (estimated at presentation) was identified in 3.5% of patients (95% CI 0.4-6.7%) and secondary bacterial infection in 14.3% of patients (95% CI 9.6-18.9%).
The overall proportion of COVID-19 patients with bacterial infection was 6.9% (95% CI 4.3-9.5%). Bacterial infection was more common in critical patients (8.1%, 95% CI 2.3-13.8%).
Peculiar species of bacterial co-pathogens were reported in 11/24 studies (45.8%), representing less than 14% of patients with reported infections.
The most common organisms reported were Mycoplasma (11 patients, 3 highlighted as M. pneumoniae), Haemophilus influenzae (5 patients), and Pseudomonas aeruginosa (5 patients).
In 14 studies the percentage of patients receiving antibiotic agents was reported. In these studies, the majority of patients received antibiotics (71.8%, CI 95% 56.1, and 87.7%).
The prescribed antibiotics were generally broad-spectrum, with fluoroquinolones and third-generation cephalosporins, which make up 74% of the prescribed antibiotics.
According to the authors, bacterial co-infection is relatively rare in patients hospitalized with COVID-19, and most of these patients may not require empirical antibacterial treatment.
 The Toronto Antimicrobial Resistance Research Network (TARRN) has prepared a specific part of its website, for the moment with non-optimal graphics, where the updates of this living review will be published.
The Toronto Antimicrobial Resistance Research Network (TARRN) has prepared a specific part of its website, for the moment with non-optimal graphics, where the updates of this living review will be published.
The low percentage of COVID-19 patients with bacterial co-infections: another synthesis of data
Another systematic review and meta-analysis Co-infections in people with COVID-19: a systematic review and meta-analysis published in August, but which included studies identified until April, highlights among the highlights:
- bacterial co-infections have been associated with mortality in previous influenza pandemics
- the percentage of COVID-19 patients with bacterial co-infection is lower than in influenza pandemics
- a higher percentage of critical patients have bacterial co-infections
- the profiles of bacterial co-pathogens are different from those of influenza co-infections
- the diagnosis of fungal co-infection is difficult, but it is strongly suspected in patients in critical conditions
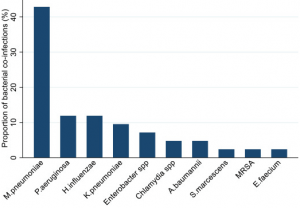
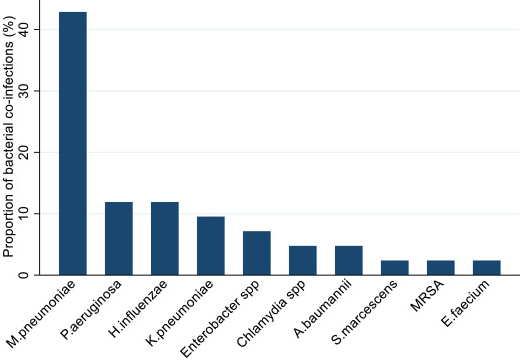
The figure shows the bacterial pathogens detected in COVID-19 patients, as a percentage (%) of the total number of detections (n = 27).
Legenda: M pneumoniae – Mycoplasma pneumoniae; P aeruginosa –Pseudomonas aeruginosa; H influenzae –Haemophilus influenzae; K pneumoniae –Klebsiella pneumoniae, A baumannii –Acinetobacter baumannii, S marcescens – Serratia marcescens, MRSA – Staphylococcus aureus resistente alla meticillina; E faecium –Enterococcus faecium.
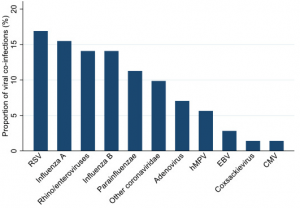
Other virus co-infections are also synthesized.
 The meta-analysis indicated that overall 7% of COVID-19 hospitalized patients had a bacterial co-infection, increasing to 14% in studies that included only intensive care patients.
The meta-analysis indicated that overall 7% of COVID-19 hospitalized patients had a bacterial co-infection, increasing to 14% in studies that included only intensive care patients.
Also in this case the authors conclude that a low proportion of COVID patients had a bacterial co-infection: lower than previous influenza pandemics.
These results do not support the routine use of antibiotics in the management of confirmed COVID-19 infection.
COVID-19 and co-infections : generalized use of antibiotics and a lurking enemy
The authors of the study “Bacterial coinfections in COVID-19: an underestimated adversary” (PDF) point out that secondary bacterial infections, although less frequent than in previous influenza pandemics, still affect some patients with COVID-19.
Mycoplasma pneumoniae, Staphylococcus aureus, Legionella pneumophila, Streptococcus pneumoniae, Haemophilus, and Klebsiella spp. are the main isolated species, which the authors cite; the co-infection with Mycobacterium tuberculosis should also be mentioned.
Bacterial co-infection rates increase in patients admitted to intensive care units and these diseases may be due to superinfections by antibiotic-resistant nosocomial bacteria.
This highlights the urgency to review the frequent and empirical prescription of broad-spectrum antibiotics in patients with COVID-19, paying more attention to evidence-based studies and respect for the principles of antimicrobial stewardship.
When two for the price of one isn’t a bargain: estimating prevalence and microbiology of bacterial co-infections in patients with COVID-19″
When two for the price of one isnt a bargain estimating prevalenceand microbiology of bacterial coinfections in patients with COVID-19Bacterial coinfections in COVID-19: an underestimated adversary
Bacterial coinfections in COVID-19Read Also:
COVID-19 In People With Down Syndrome: Mortality Up To 10 Times Higher. A Study Of The ISS
Antibiotics Resistant Bacteria: The Important Discovery Of Australia
FDA Approves Recarbio To Treat Hospital-Acquired And Ventilator-Associated Bacterial Pneumonia