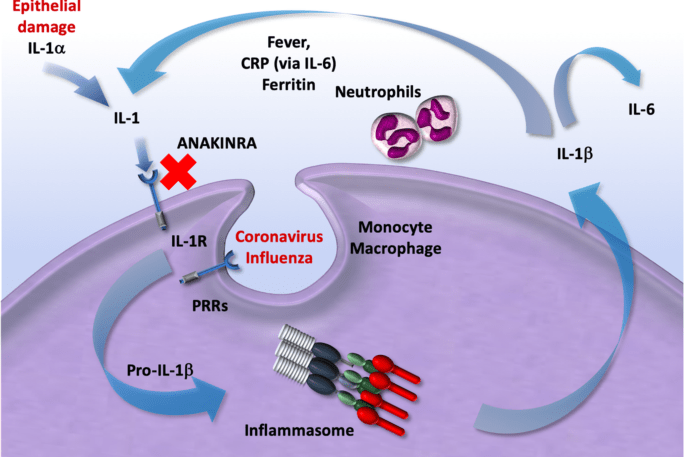
COVID-19, the role of immunomodulators (anakinra, sarilumab, siltuximab and tocilizumab) in treatment
Treatment of COVID-19 / immunomodulators such as anakinra, sarilumab, siltuximab and tocilizumab in the treatment of COVID-19: evidence of an over-activated immune response in the most severe form of COVID-19 is accumulating, with several studies exploring the therapeutic role of immunomodulation.
Systematic review and meta-analysis of data: Immunomodulators in COVID-19 treatment
Through a systematic review and meta-analysis, the study we present evaluated the efficacy of specific interleukin inhibitors for the treatment of COVID-19.
The authors of this study collected data from the Electronic databases on 7 January 2021 to identify studies of immunomodulatory agents (anakinra, sarilumab, siltuximab and tocilizumab) for the treatment of COVID-19.
The primary outcomes were severity on an Ordinal Scale measured at day 15 from intervention and days to hospital discharge.
Key secondary endpoints included overall mortality.
Results of the study on immunomodulators in COVID-19 treatment
71 studies totalling 22 058 patients were included, 6 were randomised trials. Most studies explored outcomes in patients who received tocilizumab (60/71).
In prospective studies, tocilizumab was associated with improved unadjusted survival (risk ratio 0.83, 95% CI 0.72 to 0.96, I2=0.0%), but conclusive benefit was not demonstrated for other outcomes.
In retrospective studies, tocilizumab was associated with less severe outcomes on an Ordinal Scale (generalised OR 1.34, 95% CI 1.10 to 1.64, I2=98%) and adjusted mortality risk (HR 0.52, 95% CI 0.41 to 0.66, I2=76.6%).
The mean difference in duration of hospitalisation was 0.36 days (95% CI −0.07 to 0.80, I2=93.8%).
There was substantial heterogeneity in retrospective studies, and estimates should be interpreted cautiously.
Other immunomodulatory agents showed similar effects to tocilizumab, but insufficient data precluded meta-analysis by agent.
Tocilizumab was associated with a lower relative risk of mortality in prospective studies, but effects were inconclusive for other outcomes. Current evidence for the efficacy of anakinra, siltuximab or sarilumab in COVID-19 is insufficient, with further studies urgently needed for conclusive findings.


