Lumbar puncture: what is a LP?
Lumbar puncture is a surgical procedure used to extract the cerebrospinal fluid produced by the choroid plexuses, which flows in the spinal canal of the spine, in the subarachnoid spaces and in the ventricles of the brain
LP, Lumbar puncture, is used to:
- Assess intracranial pressure and cerebrospinal fluid composition (see table Cerebrospinal fluid abnormalities in various disorders)
- Therapeutically reduce the intracranial pressure (e.g. idiopathic endocranial hypertension)
- Administer intrathecal drugs or a radiopaque contrast agent for myelography
Contraindications for LP include
- Infection at the puncture site
- Hemorrhagic diathesis
- Increased endocranial pressure secondary to an endocranial mass lesion, cerebrospinal fluid outflow obstruction (e.g., due to aqueductal stenosis or Chiari I malformation), or cerebrospinal fluid blockage in the spinal cord (e.g., due to a compressive spinal tumour)
If papilledema or focal neurological deficits are present, CT or MRI should be performed prior to lumbar puncture to exclude the presence of a mass that may cause transtentorial or cerebellar herniation.
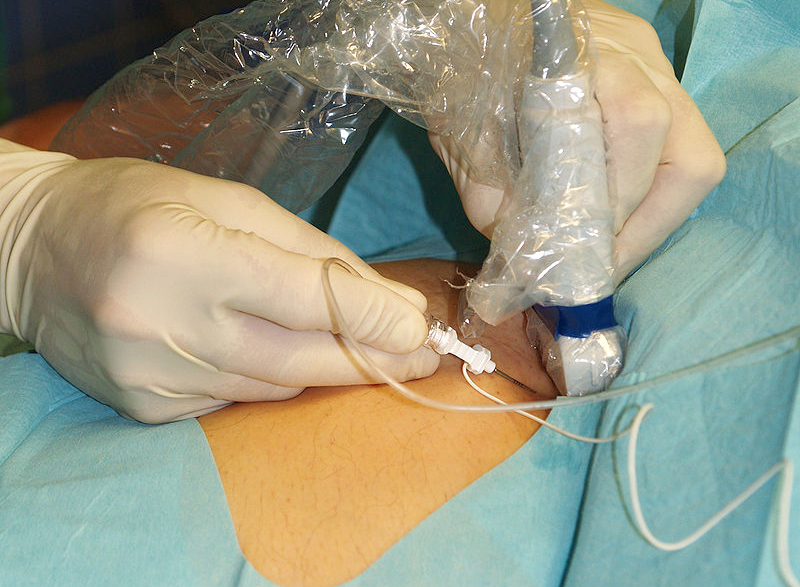
Lumbar puncture procedure
For the procedure, the patient is typically positioned in left lateral decubitus (if the physician is right-handed; to facilitate insertion of the needle into the lumbar cistern, Ed).
A cooperating patient is asked to hug the knees and lift them up (towards the chin) bent and jointed as far as possible.
Caregivers should hold patients who cannot maintain this posture, or the column can be flexed better by sitting the patient, especially obese patients, on one side of the bed and bending forward over the pull-out table beside the bed.
A 20 cm diameter area is washed with iodine tincture, then cleaned with alcohol to remove the iodine and prevent its introduction into the subarachnoid space.
A spindle-mounted spinal tap needle is inserted into the space between L3 and L4 or L4 and L5 (the spinous process of L4 is typically located on the line joining the postero-superior iliac crests); the needle is directed rostrally towards the patient’s umbilicus and always kept parallel to the floor.
Entry into the subarachnoid space is often associated with a recognisable sensation (pop); the guide is removed to allow the cerebrospinal fluid to drain.
The pressure at introduction is measured using a pressure gauge; 4 tubes are filled with approximately 2-10 mL of cerebrospinal fluid for examination.
The puncture site is then covered with a sterile adhesive plaster.
Post-lumbar puncture headache occurs in about 10% of patients.
Colour of the cerebrospinal fluid
Normal CSF is clear and colourless; ≥ 300 cells/muL results in an opaque appearance or turbidity of the CSF.
The presence of blood may be due to a traumatic puncture (pushing the needle too deep into the venous plexus along the anterior spinal canal) or a subarachnoid haemorrhage.
A traumatic puncture is distinguished by
- Gradual compensation of cerebrospinal fluid between the 1st and 4th tubes (confirmed by a decrease in the number of red blood cells)
- Absence of xanthochromia (yellowish cerebrospinal fluid due to lysed red blood cells) in a centrifuged sample
- Fresh non-knotched blood cells
In intrinsic subarachnoid haemorrhage, cerebrospinal fluid remains uniformly haematic in all tubes collected; xanthochromia is often present if several hours have elapsed since the stroke event; red blood cells are generally older and frayed in appearance.
A faintly yellow CSF may also be due to increased senile chromogens, severe jaundice, or increased protein content (> 100 mg/dL).
Number of cells and glucose and protein levels in cerebrospinal fluid
Cell counts and differential counts and glucose and protein levels are useful in the diagnostic process of many neurological diseases (see table Changes in CSF in various diseases).
Normally, the normal blood glucose to CSF ratio is about 0.6 and, except in cases of severe hypoglycaemia, the CSF is usually > 50 mg/dL (> 2.78 mmol/L).
An increase in cerebrospinal fluid protein (> 50 mg/dL) is a sensitive but non-specific index of disease; an increase in protein up to > 500 mg/dL occurs in purulent meningitis, advanced tuberculous meningitis, complete CSF blockage by spinal cord tumour or in the case of bloody lumbar puncture.
Special tests for globulins (usually < 15%), oligoclonal bands and myelin basic protein help in the diagnosis of demyelinating diseases.
Rachicentesis: staining, analysis and culture of cerebrospinal fluid
If an infection is suspected, the centrifuged CSF sediment is stained for the following:
- Bacteria (Gram stain)
- Tuberculosis (acid fast or immunofluorescent staining)
- Cryptococcus sp (Indian ink)
Larger amounts of fluid (10 mL) increase the chances of detecting the pathogen, especially acidresistant bacilli and some types of fungi, by staining and culture.
In the early phase of meningococcal meningitis or in cases of severe leukopenia, cerebrospinal fluid proteins may be too low for the bacteria to adhere to the slide during Gram staining, producing a false-negative result.
Mixing a drop of aseptic serum with the CSF sediment prevents this problem.
When haemorrhagic meningoencephalitis is suspected, a fresh preparation is used to search for amoebas.
The latex particle agglutination test and the coagglutination test may allow rapid identification of bacteria, especially when staining and culture are negative (e.g. in partially treated meningitis).
Cerebrospinal fluid should be cultured under aerobic and anaerobic conditions for acidresistant bacilli and mycetes.
Viruses are rarely isolated from cerebrospinal fluid, except for enteroviruses.
Commercially available panels are available for the identification of viral antibodies.
Venereal Disease Research Laboratories (VDRL) testing and cryptococcal antigen testing are often performed routinely.
Polymerase Chain Reaction (PCR)-based tests for herpes simplex virus and other CNS pathogens are increasingly available.
Read Also:
Ambulance Professional Back Pain War: Technology, May You Help Me?
Lumbago: What It Is And How To Treat It
Percutaneous Discectomy For Herniated Discs


