Macular degeneration: Faricimab and the new therapy for eye health
In the over 60s, the main disease that puts eye health at risk is exudative age-related macular degeneration, which in its most severe form can rapidly cause irreversible vision loss
Francesco Viola, Director of Ophthalmology at the Policlinico di Milano, is one of the authors of an article recently published in the journal ‘Lancet’ reporting promising data from a multi-centre international study in which our Ophthalmology team contributed.
This research has identified a new drug that is just as effective and safe as the current standard of care, but with the great advantage of being administered at longer intervals.
The drug has already been approved by the US Food and Drug Administration (abbreviated to FDA) and is expected to be used in Italy within the year.
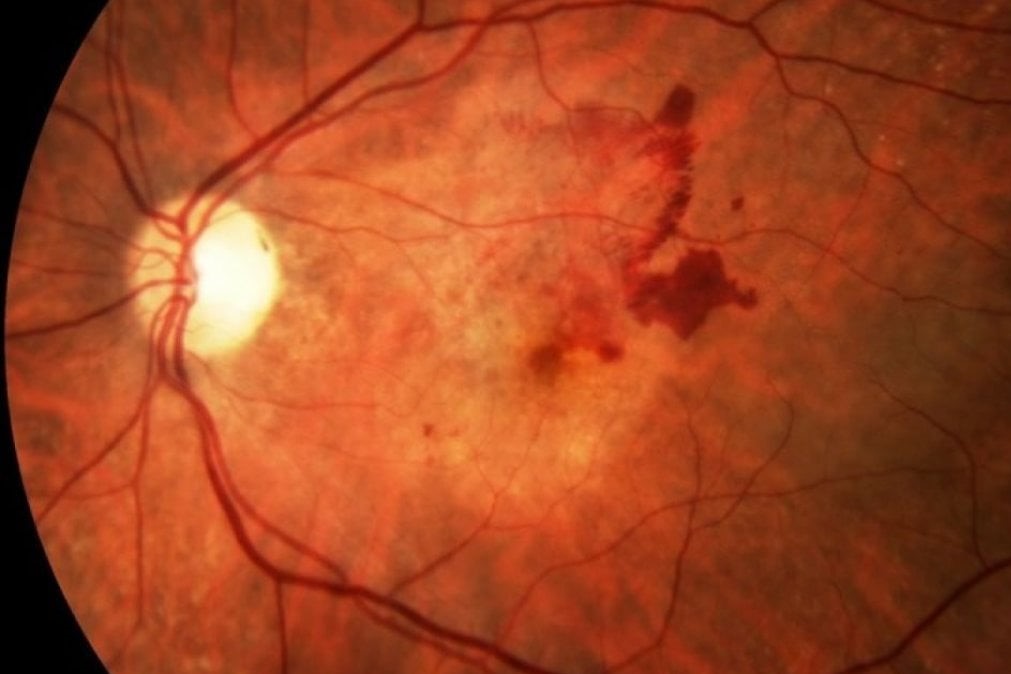
Exudative macular degeneration affects only one part of the eye: the macula
This is the central portion of the retina.
In this area, defective blood vessels form and release fluids, thus changing the internal structure of the eye.
It affects around 63,000 people in Italy each year and manifests itself in blurred vision of words when reading or a dark area in the centre of the visual field and distortion of images, compromising the quality of vision in the central part.
Current treatments block the proliferation of new abnormal blood vessels within the retina to prevent the damage from worsening, and involve injections of the drugs into the eye about every two months.
Exudative macular degeneration: New therapy involves faricimab
Faricimab is a drug that simultaneously targets two molecular targets in blood vessels: vascular endothelial growth factor (VEGF), which promotes their abnormal growth, and the protein angiopoietin (Ang-2), which contributes to their inflammation and the resulting leakage of fluid into the retina.
During the study, the treatment was well tolerated and is administered every 3-4 months in more than 75% of patients and not every 2 as the current standard of care.
“Faricimab is the first and only bispecific drug that can be injected into the eye that has been approved by the FDA.
With this therapy, patients will be able to preserve their vision by reducing the number of injections they will need to have during their lifetime.
It is an effective alternative to current therapies, with the added advantage of increasing the time between injections.
To date, around 6,000 eye injections are performed each year in our department, with the involvement of teams dedicated to these treatments.
This new therapy will have a positive impact first and foremost on patients by facilitating compliance with injections, so as to more effectively prevent blindness, but also on healthcare activities,” explained Francesco Viola, director of Ophthalmology at the Policlinico di Milano and associate professor at the University of Milan.
Read the study on exudative macular degeneration
Read Also:
Emergency Live Even More…Live: Download The New Free App Of Your Newspaper For IOS And Android
Eye Burns: What They Are, How To Treat Them
Corneal Abrasions And Foreign Bodies In The Eye: What To Do? Diagnosis And Treatment
Wound Care Guideline (Part 2) – Dressing Abrasions And Lacerations
Contusions And Lacerations Of The Eye And Eyelids: Diagnosis And Treatment
How To Irrigate The Eye And Perform Eyelid Tipping


